Role of Adenylate Cyclase 6 (AC6) in Body Phosphate Homeostasis
Funded by: The Danish Council for Independent Research | Medical Sciences Instrument (DFF – 4004-00043)
Background: Inorganic phosphate (PO43-, Pi) plays a critical role in skeletal development and bone integrity, energy metabolism, cell signaling, and regulation of protein synthesis. Pi levels in the body are balanced by dietary uptake in the small intestine and urinary excretion by the kidneys. In healthy individuals, an increase in serum Pi induces parathyroid hormone (PTH) and fibroblast growth factor 23 (FGF23) secretion. In kidney proximal tubule cells, PTH activates the Gs protein-coupled PTH 1 receptor (PTH1R) to stimulate a Gsα/AC/cAMP/PKA pathway; which subsequently reduces the expression and luminal membrane abundance of the Na-phosphate cotransporters Npt2a and Npt2c in the proximal tubules of the kidney leading to reduced renal Pi absorption and increased urinary Pi excretion (Fig. 1).
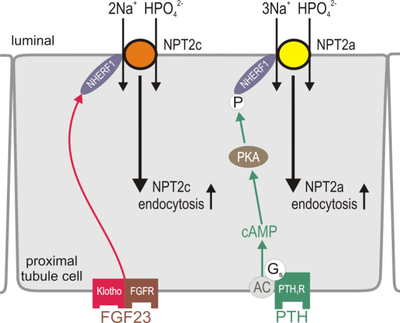
In addition a PTH/Gαq/11/PLC/IP3/Ca2+/PKC pathway has been speculated to contribute to the PTH-dependent down-regulation of Npt2a expression. Importantly, the PTH-PKC pathway is unable to compensate for the impairment of PTH-PKA signaling in patients with pseudohypoparathyroidism type Ia (PHP-Ia) or type Ib (PHP-Ib). These patients develop PTH-resistant hyperphosphatemia because of inherited mutations that abolish the activity of Gsα in the renal proximal tubule. These clinical findings indicate a dominant role for the PTH-PKA pathway in the PTH-dependent regulation of Npt2a expression and urinary Pi excretion. PTH also increases 1α-hydroxylase secretion by the kidney, which increases 1,25-dihydroxyvitamin D3 production and enhances intestinal Pi absorption through Npt2b. A balance between these mechanisms allows plasma Pi levels to remain within a narrow range to maintain physiological processes (Fig. 2).
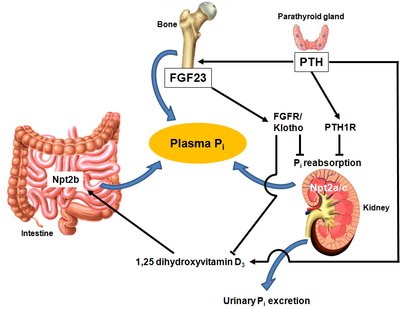
Problem statement and objectives: Critical to maintaining body Pi balance are the effects of PTH, which are mediated via a Gsα/AC/cAMP/PKA pathway. However, which AC isoform(s) mediate PTH-induced effects, either directly or indirectly, remains unknown. This proposal will determine the role of AC6 in body Pi homeostasis using various AC6 gene knockout mice. Furthermore, we hypothesize that altered sensitivity to PTH in the kidneys of AC6 gene modified mice will lead to changes in FGF23 and 1,25-dihydroxyvitamin D3 levels and thus indirect alterations in intestinal Pi transport and bone mineralization. The research is broken down into 3 research objectives:
1) Objective I will assess the role of AC6 in renal Pi transport and molecular pathways of Pi uptake
2) Objective II will assess Pi uptake in the intestine and if changes occur secondary to renal effects
3) Objective III will study the effects of altered Pi homeostasis on bone morphology/mineralization
Perspectives for the field: The experiments outlined will employ a novel, mostly in vivo, approach to determine the role of AC6 in Pi homeostasis. Our studies will provide mechanistic insight into the role of AC6 in Pi handling by the kidneys (including a transport profile along the proximal tubule), intestinal Pi transport, and bone mineralization – which combined makes our studies of high importance for various fields. We will generate two novel mutant mouse models to decipher the contribution of AC6 for renal versus intestinal Pi homeostasis that will provide unprecedented insights into the regulation of Pi homeostasis. Currently, no specific protein involved in Pi homeostasis has become a pharmacological target for activation and/or blockade making it necessary to study basic physiological questions to provide new targets for clinical exploitation.